 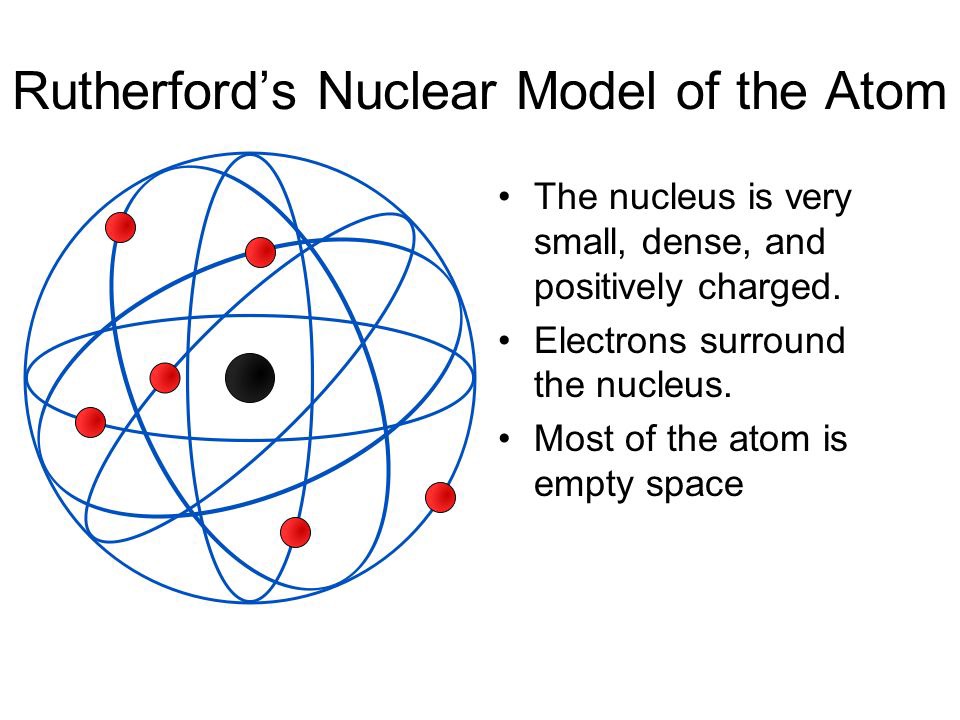 
In 1808, he presented his atomic theory which was a turning point in the study of matter.Dalton’s atomic theory provided an explanation for the law of conservation of mass and the law of definite proportions.His theory was based on the laws of chemical combination. He took the name ‘atoms’ as given by the greeks and said that the smallest particles of matter are atoms.British chemist John Dalton provided the basic theory about the nature of matter.The problem faced by scientists was to give appropriate explanations to the above laws.This law was stated by Proust as “in a chemical substance the elements are always present in definite proportions by mass”. Law of constant proportions is also known as the law of definite proportions.Thus, if 9 g of water is decomposed, 1 g of hydrogen and 8 g of oxygen are always obtained. In a compound such as water, the ratio of the mass of hydrogen to the mass of oxygen is always 1:8.Law of constant proportions: Lavoisier, along with other scientists, noted that many compounds were composed of two or more elements and each such compound had the same elements in the same proportions, irrespective of where the compound came from or who prepared it.Law of conservation of mass: Law of conservation of mass states that mass can neither be created nor destroyed in a chemical reaction.Atoms of the same element or of different elements can join together to form molecules.A molecule can be defined as the smallest particle of an element or a compound that is capable of an independent existence and shows all the properties of that substance.A molecule is in general a group of two or more atoms that are chemically bonded together, that is, tightly held together by attractive forces.These molecules or ions aggregate in large numbers to form the matter that we can see, feel or touch. Atoms of most elements are not able to exist independently.Source | Credits | Picture Credits: NCERT General Science. 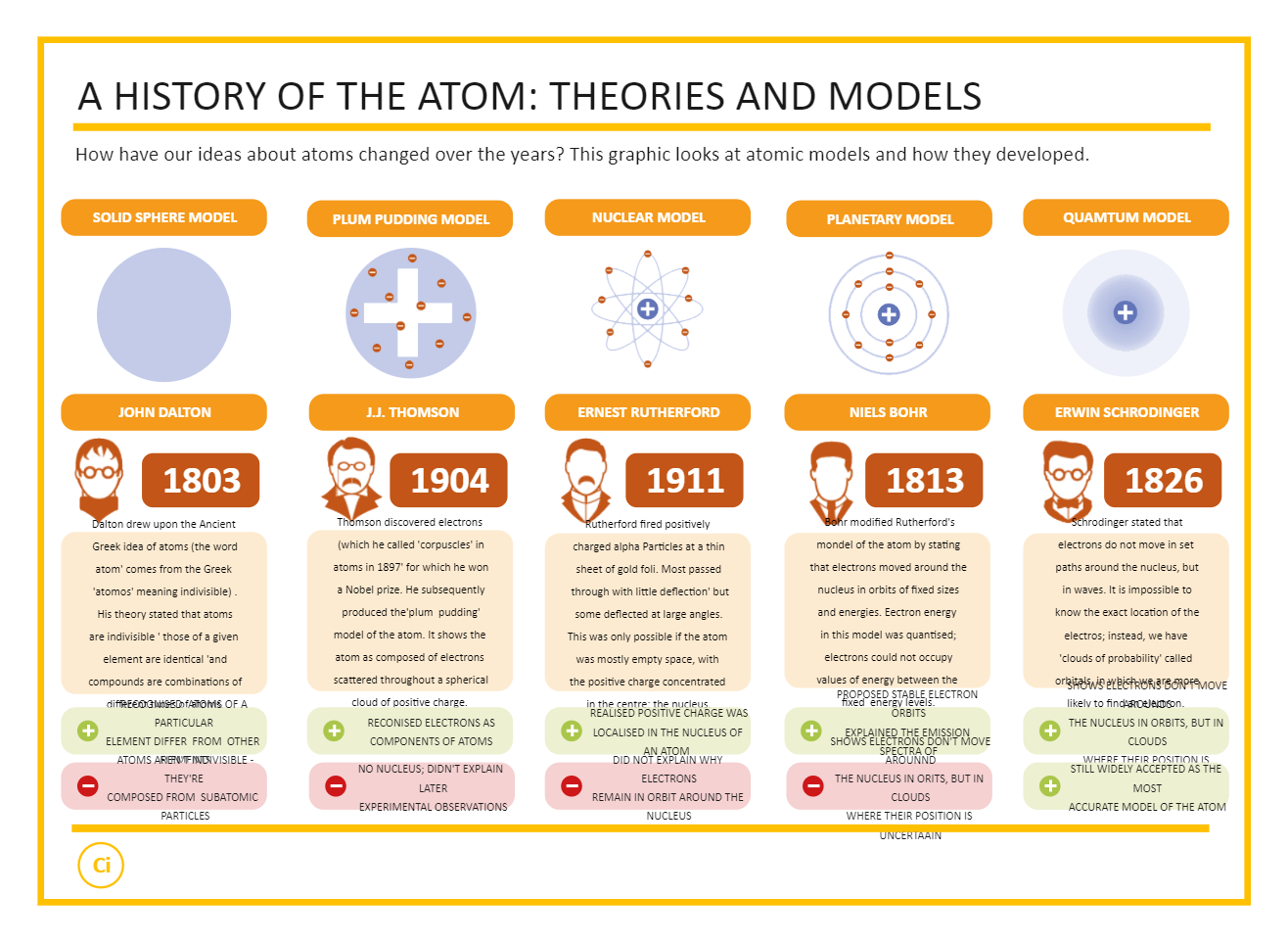
Atoms Molecules, John Dalton’s Atomic Theory, Atomic, Molecular mass, Mole concept, Structure of The Atom, Valency, Atomic Number, Mass Number, Isotopes. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
